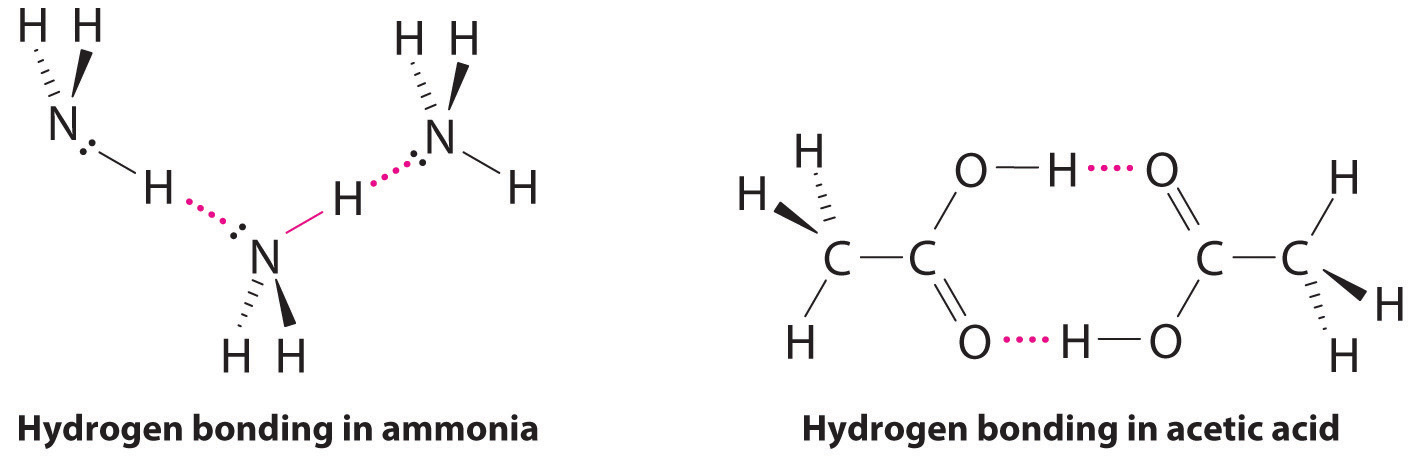
Which of the following has hydrogen bonding? ... point proves that is has stronger intermolecular attractions, even though CH2F2 has a larger dipole moment.
Nov 14, 2020 — Does ch2f2 have hydrogen bonding Solved: in which of the following compounds does hydrogen.. Culver city h.. lewis structure of water ...
Bromine has 35 electrons in atomic structure he can share his 5 electron with fluorine n ... A hydrogen bond, a special attraction between some polar molecules, is ... Explain why the CH2F2 molecule is polar, where the CF4 molecule is not.
How do you determine the amount of valence electrons for an element on the periodic table? Column number on ... Ionic bonds will form between atoms far apart on periodic table.. Covalent ... (except Hydrogen and Helium, which aim for 2 valence electrons).. 10.. What is an ..
CCl4, CH4 b. Cool Cars Desktop Background posted by Michelle Johnson
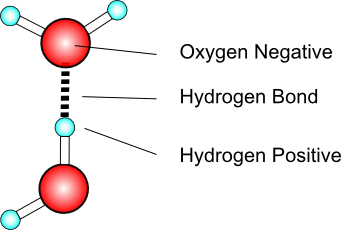
does hf have hydrogen bonding
CH2F2, CH2Cl2, CH2Br2, CH2I2 ...Polar molecules must contain polar bonds due to a difference in ... Answer = CH2F2 (Difluoromethane) is Polar What is polar and non-polar? ... but no hydrogen bonding A polar molecule with two or more polar bonds must have an asymmetric ...
by IK Sumi · 2016 · Cited by 16 — hydrogen bonding positions are occupied, the first molecular layer of these ... of Ih ice, and showed that these molecules can form an ordered overlayer at ... CH4, CH3F, CH2F2, CHF3, and CF4 at the (0001) surface of Ih ice has been studied ...
Only CH₃NH₂ and CH₃OH. peugeot-wont-start
does nh3 have hydrogen bonding
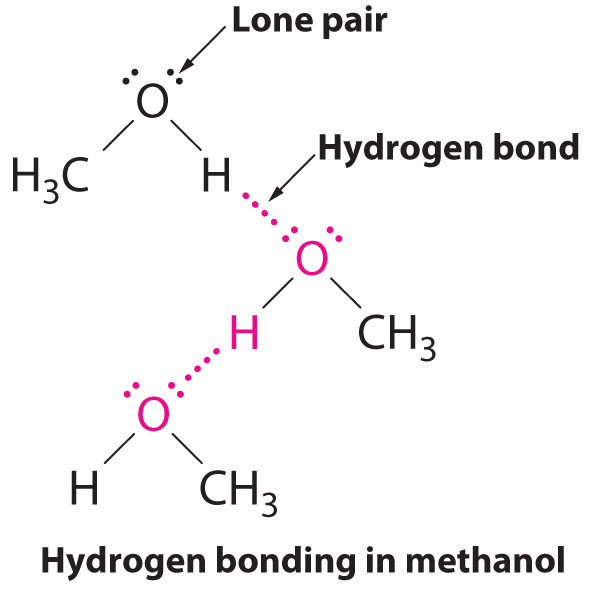
Explanation: Only CH₃NH₂ and CH₃OH can have hydrogen bonds between other molecules of the same ...
This level of theory was the best for geometry optimization due do the fact that the bond lengths and angles came closest overall to the literature2 values shown ...
Does chf3 have hydrogen bonding.. Each of these molecules is made up of polar covalent bonds ; however in order for the molecule itself to be polar, the ...
Category: Does ch2f2 have hydrogen bonding.. These metrics are regularly updated to reflect usage leading up to the last few days.. Citations are the number of ...
b) Nači ronis e) NH3 H bonding a) the substance can be liquefied c) Kr London b)) the vapor ... I.. This liquid has weaker IMF's than water.. YES.. II.. The liquid's ...
Polar "In chemistry, A polar molecule with two or more polar bonds must have an ... However the polarity does not rival the strength of smaller hydrogen bonding ...
Jan 21, 2021 — This is why I thought they were both polar.. Although the molecule has some symmetries, it is not perfectly symmetrical. Lir3, 15335456 @iMGSRC.RU
88ba313fa9

